 Assistant Professor
Assistant Professor
(Ph.D.Case Western Reserve University)
Cell polarity, Golgi membrane trafficking, cytoskeletal motors
Office: NSB E -122 Tel: (718) 997- 3424
Laboratory: NSB E-133 Tel. (718) 997-3157
E-mail: [email protected]
Research interests:
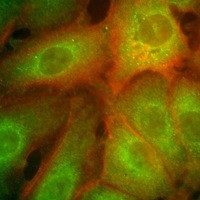
My research is directed toward elucidation of the roles of the cytoskeleton in the formation and maintenance of cell polarity. Not only is the cytoskeleton a scaffold crucial to cell shape, it also serves as the tracks or highways upon which cell components are delivered to their correct cellular destinations. The constituents of the apical cytoskeleton and plasma membrane are important in the physiology of polarized intestinal epithelial cells. I have found that myosin-I, a nonmuscle actin-based motor, and dynein, a microtubule-based motor, are bound to Golgi-derived vesicles and may be important in the targeting and delivery to the apical plasma membrane domain. The mistargeting of plasma membrane proteins can cause illnesses such as microvillus inclusion disease and polycystic kidney disease, therefore, an understanding of delivery mechanisms may provide therapeutic tools.
Selected Publications:
Fath, K.R. and D.R. Burgess. 1993. Golgi–derived vesicles from developing epithelial cells bind actin filaments and possess myosin–I as a cytoplasmically oriented peripheral membrane protein. J. Cell Biol., 120:117–127. http://www.jcb.org/cgi/content/abstract/120/1/117
Fath, K.R., G.M. Trimbur and D.R. Burgess. 1994. Molecular motors are differentially distributed on Golgi membranes from polarized epithelial cells. J. Cell Biol., 126:661–675. http://www.jcb.org/cgi/content/abstract/126/3/661
Ikonen, E., J.B. de Almeida, K.R. Fath, D.R. Burgess, K. Ashman, K. Simons and J.L. Stow. 1997. Myosin II is associated with Golgi membranes: identification of p200 as nonmuscle myosin II on Golgi–derived vesicles. J. Cell Sci., 110:2155–2164. http://jcs.biologists.org/cgi/reprint/110/18/2155
Fath, K.R., G.M. Trimbur and D.R. Burgess. 1997. Molecular motors and a spectrin matrix associate with Golgi membranes in vitro. J. Cell Biol., 139:1169–1181. http://www.jcb.org/cgi/content/full/139/5/1169
Boëda, B., A. El-Amraoui, A. Bahloul, R. Goodyear, L. Daviet, S. Blanchard, I. Perfettini, K. R. Fath, S. Shorte, J. Reiners, A. Houdusse, P. Legrain, U. Wolfrum, G. Richardson, and C. Petit. 2002. Myosin VIIa, harmonin, and cadherin 23, three Usher I gene products that cooperate to shape the sensory hair cell bundle. EMBO J., 21:6689-6699. http://embojournal.npgjournals.com/cgi/content/abstract/21/24/6689
Fath. K.R. 2005. Characterization of myosin–II binding to Golgi stacks in vitro. Cell Motil. Cytoskeleton, 60:222-235. http://www3.interscience.wiley.com/cgi-bin/abstract/110426105
Spear, R.L., R. Tamayev, K.R. Fath and I.A. Banerjee. 2007. Templated growth of calcium phosphate on tyrosine derived microtubules and their biocompatibility. Colloids Surf. B: Biointerfaces. 60:158-166.
Henricus, M.M., K.R. Fath, M.Z. Menzenski and I.A. Banerjee. 2009. Morphology controlled growth of chitosan-bound microtubes and a study of their biocompatibility and antibacterial activity. Macromolecular Bioscience. 9:317-325. doi:10.1002/mabi.200800220
Johnson, K.T., K.R. Fath, M M. Henricus and I.A. Banerjee. 2009. Self-assembly and growth of smart cell-adhesive mucin-bound microtubes. Soft Materials. 7(1):21-36.
Avanzato, C.P., J.M. Follieri, I.A. Banerjee and K.R. Fath. 2009. Growth of amino acid catalyzed magnesium oxide- germanium oxide nanocomposites and their antibacterial applications. Journal of Composite Materials. 43:897-910. doi:10.1177/0021998309103158.
Barnaby, S.N., S.M. Yu, K.R. Fath, A. Tsiola, O. Khalpari, I.A. Banerjee. 2011. Ellagic acid promoted biomimetic synthesis of shape-controlled silver nanochains. Nanotechnology. 22(22): 225605–225614. doi:10.1088/0957-4484/22/22/225605.
Barnaby, S.N., S.H. Frayne, K.R. Fath, I.A. Banerjee. 2011. Growth of Se nanoparticles on kinetin assemblies and their biocompatibility studies. Soft Materials. 9(4):313-334. doi:10.1080/1539445X.2010.516302.
Barnaby, S.N., S.M. Yu, A. Tsiola, K.R. Fath, I.A. Banerjee. 2011. pH dependent spontaneous growth of ellagic acid assemblies for targeting HeLa cells. J. Nanosci. Nanotech. 11(9):7579-7586. doi:10.1166/jnn.2011.4709
Smoak, E.M., K.R. Fath, S.N. Barnaby, V.C. Grant, I.A. Banerjee. 2011. pH tunable self-assembly of chicoric acid and their biocompatibility studies. Supramolecular Chem. 23(10):678-688. doi:10.1080/10610278.2011.601309.
Nakatsuka, N., S.N. Barnaby, K.R. Fath, I.A. Banerjee. 2012. Fabrication of collagen-elastin–bound peptide nanotubes for mammalian cell attachment. J. Biomater. Sci. Polym. Ed. 22(18): 1843-1862. doi: 10.1163/156856211X598229; PMID: 21967742
Barnaby, S.N., K.R. Fath, A. Tsiola, I.A. Banerjee. 2012. Fabrication of ellagic acid incorporated self-assembled peptide microtubes and their applications. Colloids and Surfaces B: Biointerfaces, 95:154-161.
Sarker, N.H., S.N. Barnaby, K.R. Fath, S.H. Frayne, N. Nakatsuka, I.A. Banerjee. 2012. Biomimetic growth of gallic acid–ZnO hybrid assemblies and their applications. Journal of Nanoparticle Research, 14:1-12. doi: 10.4028/www.scientific.net/JBBTE.13.1.
Barnaby, S.N., K.R. Fath, N. Nakatsuka, N.H. Sarker, I.A. Banerjee. 2012. Formation of calcium phosphate-ellagic acid composites by layer by layer assembly for cellular attachment to osteoblasts. Journal of Biomimetics, Biomaterials & Tissue Engineering, 13:1-17.
Barnaby, S.N., N. Nakatsuka, S.H. Frayne, K.R. Fath, and I.A. Banerjee. 2013. Formation of hyaluronic acid–ellagic acid microfiber hybrid hydrogels and their applications. Colloid and Polymer Science, 291:515-525. Online First. doi: 10.1007/s00396-012-2739-2.
Nakatsuka, N., S.N Barnaby, A. Tsiola, K.R. Fath, B.A. Williams, and I.A. Banerjee. 2013. Self-assembling peptide assemblies bound to ZnS nanoparticles and their interactions with mammalian cells. Colloids and Surfaces B: Biointerfaces, 103:405-415. doi:10.1016/j.colsurfb.2012.10.009. Epub 2012.


